Accenture creates a regulatory document authoring solution using AWS generative AI services
AWS Machine Learning
FEBRUARY 6, 2024
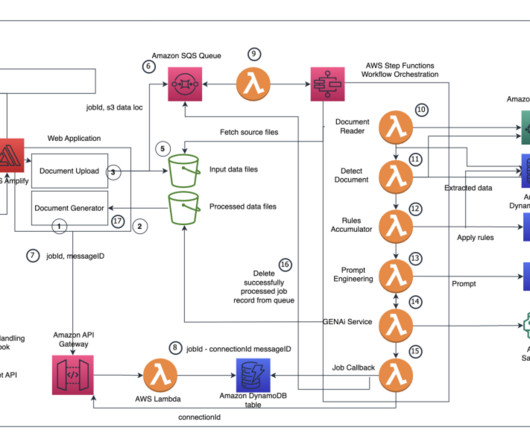
A key part of the submission process is authoring regulatory documents like the Common Technical Document (CTD), a comprehensive standard formatted document for submitting applications, amendments, supplements, and reports to the FDA. This revolutionary approach compresses the time and effort spent on CTD authoring.















































Let's personalize your content